[Please refer to the instruction manual.]
1. General Name
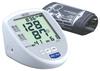
Automatic Electronic Blood Pressure Monitor
2. Product Name
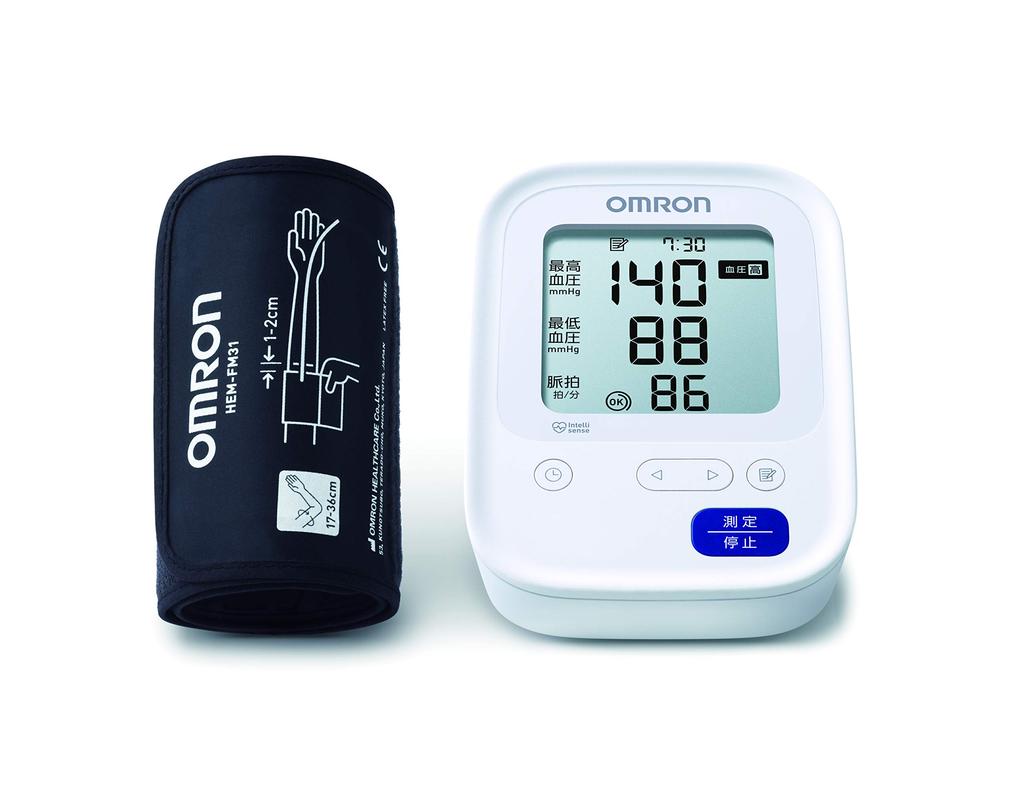
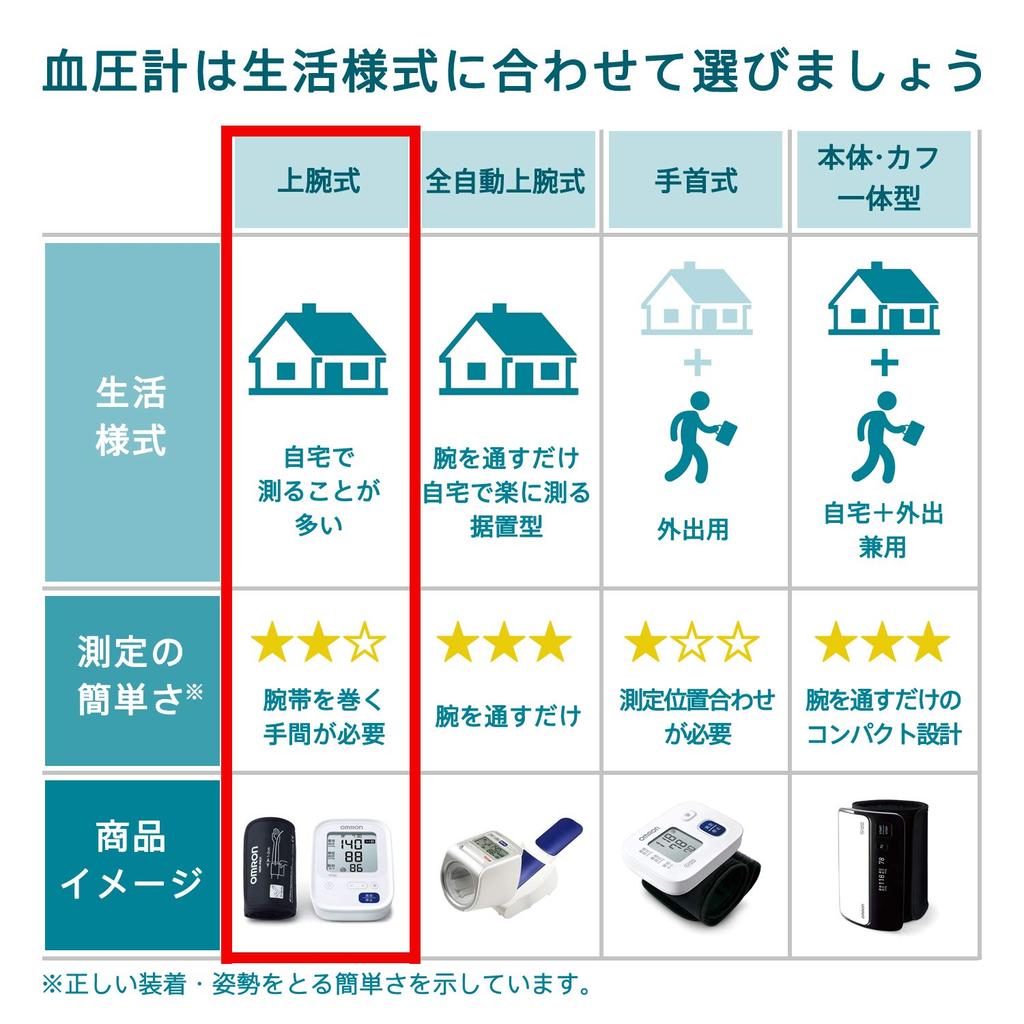
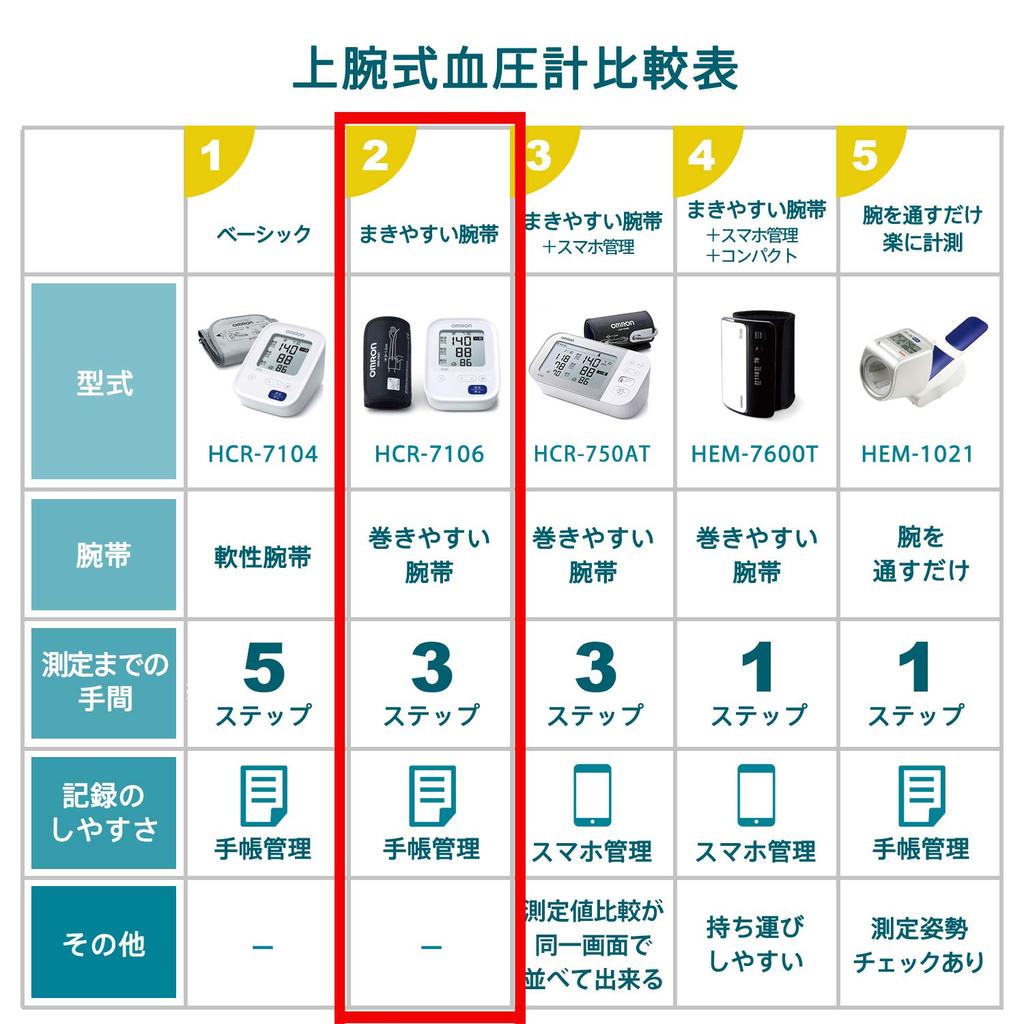
Omron Upper Arm Blood Pressure Monitor Standard 19 Series
3. Contraindications/Prohibitions
(1) Self-diagnosis of measurement results or treatment based on self-diagnosis
[This may lead to worsening of symptoms.]
(2) Measurement on an injured or injured arm
[This may lead to worsening of symptoms.]
(3) Measurement on an arm receiving intravenous infusion or blood transfusion
[This may lead to worsening of symptoms.]
(4) Use near flammable or combustible gases
[This may cause ignition, fire, or explosion.]
(5) Use beyond the service life
[This may result in inaccurate measurements.]
(6) Use by an unspecified number of people in medical institutions or public places
[This may cause accidents or problems.] ]
4. Item Specifications
External Dimensions: Approx. 105 (W) x 87 (H) x 153 (D) mm
Weight: Approx.
310 g (excluding batteries)
Applicable Arm Circumference: 17 ~ 36 cm
Operating Environment Conditions: +10 ~ +40 ℃ / 15 ~ 90% RH (non-condensing) / 800 ~ 1060 hPa
5. Precautions for Use
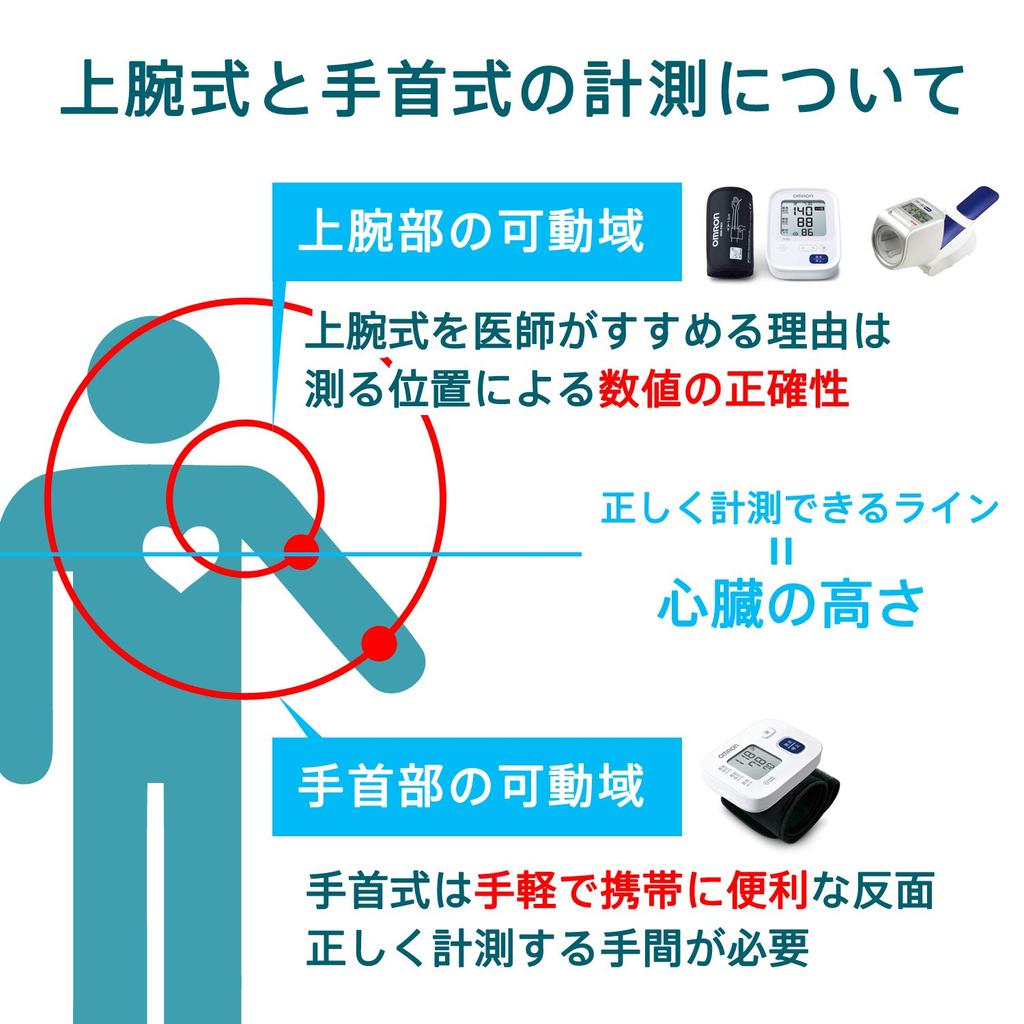
1. Individuals with severe circulatory disorders or blood diseases should use this device under the guidance of a physician.
2. If an abnormality occurs, remove the cuff from the arm.
3. Do not use on infants or individuals unable to express their wishes.
4. Do not inflate the cuff above 300 mmHg.
5. Do not use for purposes other than blood pressure measurement.
6. Do not use cuffs other than the dedicated cuff.
7. Do not use mobile phones or smartphones near the blood pressure monitor.
8. Do not disassemble or modify the main unit or the dedicated cuff.
9. Insert the battery with the correct polarity (+/-).
10. Use the specified battery.
11. Remove the battery when not using the device for an extended period.
6. Manufacturer and Distributor Information (Contact information for product inquiries is clearly stated)
Distributor: Omron Healthcare Co., Ltd.
Phone: 0120-30-6606
(Omron Customer Service Center)
7. Intended Use or Effect
To non-invasively measure systolic and diastolic blood pressure for health management.
Medical Device Classification: Specified Controlled Medical Device
Medical Device Certification (Approval) Number: 301AABZX00022000